5/22/14
There are several etiologies for limbal stem cell deficiency of the front of the eye. These include chemical and thermal burns, Steven-Johnson syndrome (which is an autoimmune severe allergic reaction that causes a burn from within), congenital aniridia, and a few other insults such as contact lens over-wear. All of these cause severe ocular surface scarring and problems with the cornea. Many eyes with these diseases have problems with corneal healing. They do not have the stem cells to support ocular surface health. The scarring can be so severe in many cases that severe corneal blindness can result.

In these cases, a simple corneal transplant will quickly fail and not result in any visual improvement. The reason for this is that the stem cells of the ocular surface have been damaged or burned out.
Visual rehabilitation for these eyes usually requires a limbal-corneal stem cell transplantation. The stem cells can be taken from the other healthy eye of the same patient, a living related donor, and or cadaveric tissue. In most cases systemic immunosuppression medications need to be taken for 1 to 3 years following surgery in order to minimize risk of rejection. Management of these patients is done in conjunction with an immunologist or a transplant specialist who can co-manage and monitor for systemic toxicity while the patient is on the these immunosuppressive medications. As most of these eyes also have concomitant glaucoma and scarring of the eyelids to the globe, co-management with a glaucoma specialist and an oculoplastic specialist is also required.
For patients who cannot be on systemic immunosuppression for other health reasons such as diabetes or cancer, they may require an artificial corneal transplantation. The artificial corneal transplantation is reserved as a last step for visual rehabilitation in these eyes. The only artificial cornea that has shown potential, is the Boston keratoprosthesis. Even this artificial cornea carries a high risk for infection and glaucoma. Very close monitoring of eyes that have an artificial cornea is required to monitor for infection and glaucoma progression. However these eyes do not require systemic immunosuppression.
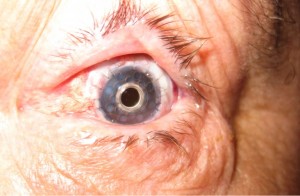
The management of eyes with severe ocular surface disease is a difficult one for the cornea specialist. A subspecialist in severe ocular surface disease and limbal stem cell transplantation is required to manage these very sick eyes. At the Gavin Herbert Eye Institute, we have developed a team approach for the management of severe ocular surface disease patients and have successfully treated and are managing many patients who have otherwise no place to go.
 Marjan Farid, MD
Marjan Farid, MD
Director of Cornea, Cataract, and Refractive Surgery
Vice-Chair of Ophthalmic Faculty
Director of the Cornea Fellowship Program
Associate Professor of Ophthalmology
Gavin Herbert Eye Institute, University of California, Irvine



