Research on mitochondrial DNA shows promise for treating AMD
For the past few years, DEF Research Director Dr. M. Cristina Kenney has been researching the relationship of mitochondria and age-related macular degeneration (AMD). She found that damaged mitochondria from people with AMD send signals that can cause retinal cells to die at an increased rate, compared with people who had healthy mitochondria and no AMD. That research led to the exploration of stimulating mitochondria to support retinal cell health in an effort to retain or restore vision for people with AMD.
Mitochondria in Cells
Cells are the basic building blocks of all living things. The human body is composed of trillions of cells. They provide structure for the body, take in nutrients from food, convert those nutrients into energy and carry out specialized functions. Cells also contain the body’s hereditary material (DNA) and so they can make copies of themselves.
Mitochondria are tiny structures inside cells whose function is to produce energy, like a battery in a flashlight, to keep cells alive. Each cell contains hundreds to thousands of mitochondria, which are located in the fluid that surrounds the nucleus. Although most DNA is packaged in chromosomes within the nucleus of a cell (nuclear DNA), mitochondria also have a small amount of their own DNA, known as mitochondrial DNA or mtDNA.
Because only egg cells contribute mitochondria to a developing embryo, only females can pass on the mitochondrial DNA to their children.
Mitochondrial Haplogroups
The mtDNA can be classified into categories called haplogroups, which represent different ancient, geographically separated groups of people. For example, African-Americans and people of ancient African lineage have inherited L haplogroup mitochondria from their mothers no matter where they currently live.

Similarly, most Ashkenazi Jewish populations (primarily those Jews whose families originated in Eastern or Central Europe) possess mitochondria of the K haplogroup. People with this haplogroup of mtDNA seem to be susceptible to a variety of age-related diseases, including age-related macular degeneration (AMD).
The incidence of AMD varies a lot among different ethnic/racial populations. For example, in the United States, the likelihood of losing vision from AMD is very low for a person with an African maternal background but it is much higher in people of European descent. Similarly, in an Israeli eye clinic, of the people who had AMD, 96% were Jewish while only 4% were of Arab descent. This suggests that European mtDNA in retinal cells of Caucasians may be the reason they are more susceptible to AMD.
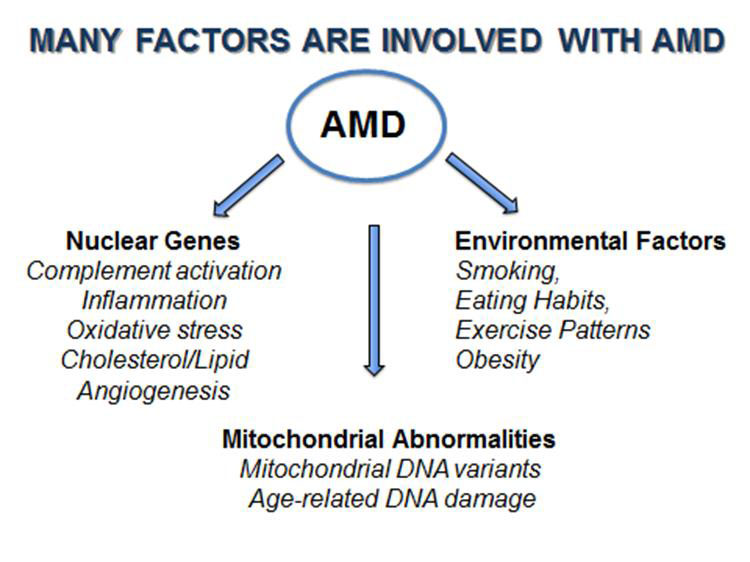
It has been recognized that AMD is a very complex disease with many factors involved (Figure 1). There are more than 30 genes associated with AMD, representing many different biological pathways. In addition, mitochondrial damage and specific mtDNA haplogroups have been associated with AMD. Finally, it is recognized that environmental factors, such as smoking and obesity, increase the risk to develop AMD.
Although millions of dollars and thousands of man-hours have been invested in finding the causes and treatments for AMD, we still do not understand how to prevent the most common form of AMD. One major difficulty has been that when we study a diverse group of individuals, each with hundreds of different nuclear and mitochondrial genes, it is very difficult to identify the causes and pathways involved with developing AMD and determining effective treatments. One drug may not help everyone and different people develop different types and severities of AMD.

Kenney’s approach to this dilemma has been to SIMPLIFY THE TESTING SYSTEM (Figure 2). In her research with different ethnic/racial groups, Kenney has found that the Ashkenazi Jewish population (K haplogroup) is an excellent group in which to study age-related diseases. This group has very well characterized nuclear and mitochondrial genes, the population tends to relatively homogenous and to marry within their community. Finally, the Ashkenazi Jewish population has longevity, which increases the likelihood that they will develop aging diseases, such as AMD.
Kenney’s laboratory has created a “cybrid” test system, which are cell lines with identical nuclei and nuclear DNA, but different mitochondrial DNA so that the differences in the cell behavior can be attributed to the different mitochondrial DNA (see the following cybrid story on 11/17/15). Using the cybrid system, Kenney has compared cell behavior of mitochondria from subjects with the K (Ashkenazi Jewish) haplogroups and the mitochondria from people of the H haplogroup (Figure 3), the most common European haplogroup.
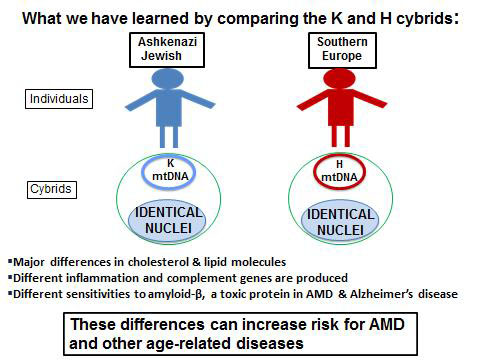
There are:
- Major differences in production of cholesterol and lipid molecules
- Altered levels of inflammation
- Differences in their responses to toxic effects of amyloid-? (a toxic protein associated with AMD and Alzheimer’s disease)
These differences are important contributors to AMD and other age-related diseases.
Significance of the Findings
Maternally inherited mitochondrial DNA can influence how a person’s cells respond to stress and this can contribute to age-related diseases. This is a completely new way of thinking about common aging diseases and offers new approaches to treatment and prevention of those diseases.
Future Studies
Kenney’s laboratory will continue to use the K haplogroup cybrid model to study the mitochondrial DNA, with the goal of blocking the harmful events that cause early retinal cell death, such as that seen in AMD. An additional advantage of cybrids is that they are unique to the donor whose blood was used to make them. Therefore with these “personalized cybrids,” Kenney can test the responses of the personalized cybrids to drugs that are currently being used for AMD (Lucentis™, Avastin™ and Eylea™). They can also be used to identify novel, new drugs that can protect the cells from early cellular death, a major event in the dry form of AMD. This research shows great promise in developing personalized treatments for AMD and other age-related diseases.
11/12/15
 Anthony B. Nesburn, MD FACS
Anthony B. Nesburn, MD FACS
President/Medical Director
Discovery Eye Foundation

